Near-to-target-aware OARs segmentation in cervical HDR brachytherapy via deep learning
OC-0132
Abstract
Near-to-target-aware OARs segmentation in cervical HDR brachytherapy via deep learning
Authors: Ruiyan Ni1, Benjamin Haibe-Kains1,2, Alexandra Rink1,2,3,4
1University of Toronto, Department of Medical Biophysics, Toronto, Canada; 2University Health Network, Princess Margaret Cancer Center, Toronto, Canada; 3University of Toronto, Department of Radiation Oncology, Toronto, Canada; 4University Health Network, TECHNA Institute, Toronto, Canada
Show Affiliations
Hide Affiliations
Purpose or Objective
Deep learning (DL) has been used to automate and speed up the time-consuming manual contouring step for organs and targets in radiotherapy. In cervical high dose-rate brachytherapy (HDR-BT), the delineation of OARs far from the target is not considered during treatment planning because treatment planning is driven by OAR dose to 2 cubic centimeters (D2cm³) closest to the target. Geometrics in distal OAR segmentation does not necessarily translate to dosimetric or clinical impact. A novel loss function was developed in this study to guide the network’s attention and ensure highly accurate OAR segmentation close to the applicator which will increase the clinical relevance and acceptability of the model’s predictions.
Material and Methods
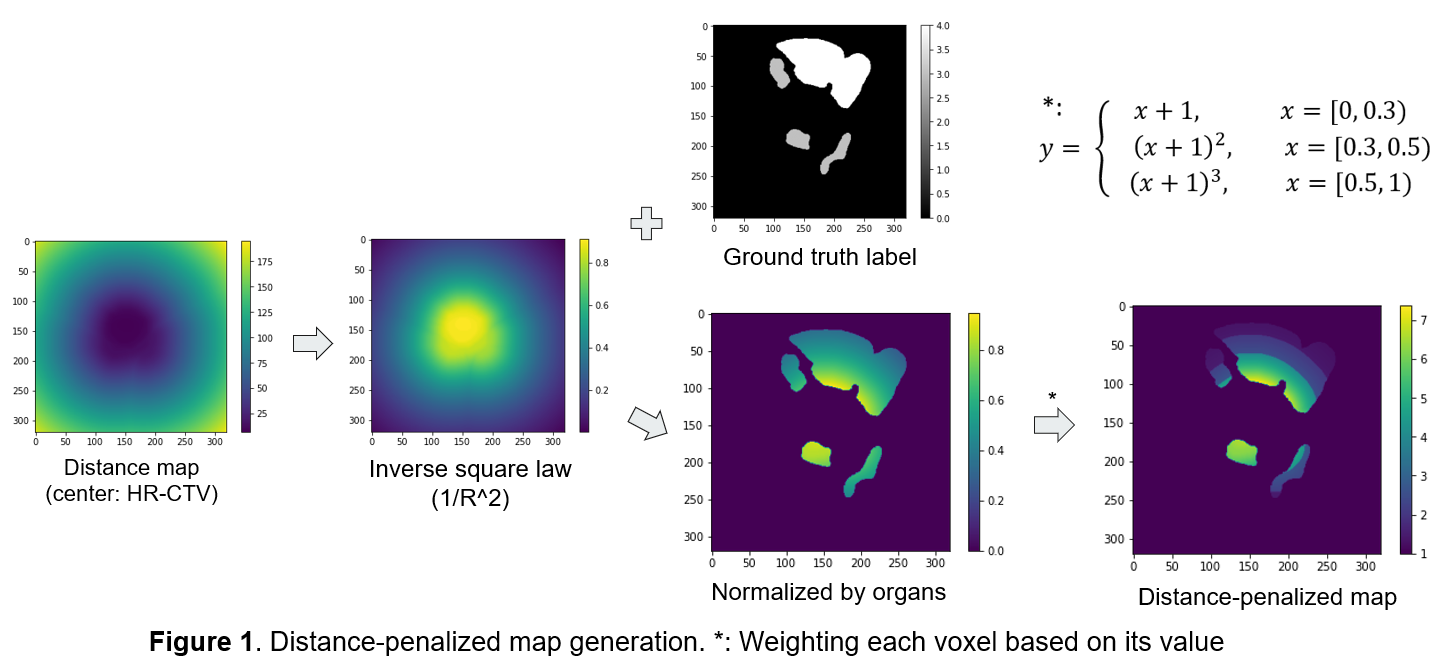
A dataset of 130 T2-weighted MR images with clinically used contours was built from 39 cervical cancer patients undergoing HDR-BT. Four OARs (bladder, rectum, sigmoid, and small bowel) were segmented. The distance-penalized (DP) map was calculated by (1) generating a distance transform map with HR-CTV as the center; (2) applying inverse square law and normalizing by each organ (range: [0,1]); (3) adding one to the map and weighting it by values, as shown in Figure 1. The generated map D was used to penalize the training errors. The training goal is to minimize the penalized multi-class combo loss L in Equation (1), where L_DPCE and L_DPDice are the DP cross-entropy loss and DP Dice loss; N and C are the numbers of voxels and classes. The network was trained on 119 cases (35 patients) with 5-fold cross-validation via 3D U-Net. Combo loss without DP (L_CE + L_Dice) was used as the baseline for comparison. The segmentation performance was evaluated by the volumetric Dice Similarity Coefficient (DSC) and a newly proposed weighted DSC (wDSC) as shown in Equation (2), where A, B and dist are the ground truth, prediction and penalty term for each class, respectively.
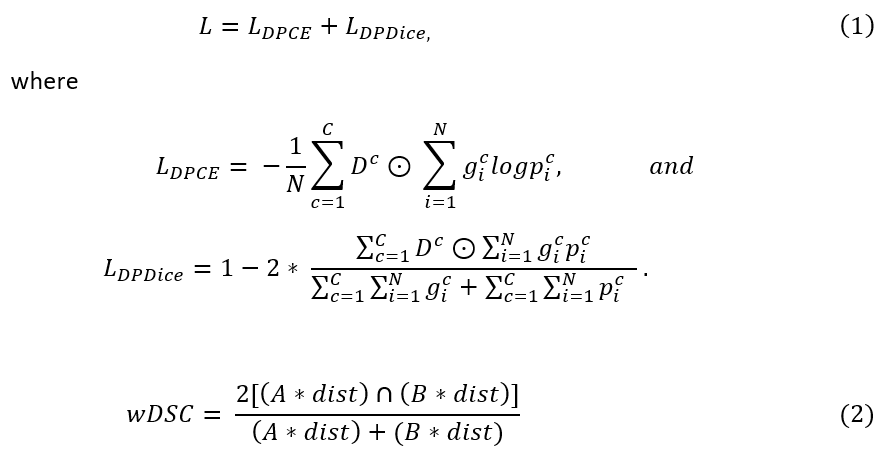
Results
The DSCs (mean±standard deviation) for the 11 test cases were 0.93±0.05 (bladder), 0.85±0.08 (rectum), 0.79±0.10 (sigmoid), and 0.73±0.17 (small bowel) with the proposed loss function, which was similar to the baseline results (mean DSCs diff <0.02). The mean wDSCs were 0.98 and 0.98 for bladder, 0.93 and 0.92 for rectum, 0.91 and 0.89 for sigmoid, and 0.87 and 0.84 for small bowel in our proposed loss and baseline loss, respectively, indicating that our method has significant improvement in near-to-target region segmentation, especially for poorly predicted organs, like sigmoid and small bowel.
Conclusion
We have proposed a novel loss function for guiding the network to highly accurate proximal OARs segmentation. This subregion-aware loss function has shown promising results in improving the performance of near-to-target OAR regions and maintaining the whole organ segmentation performance, which addressed the real clinical needs of OAR contouring and will help to further generate optimized treatment plans.